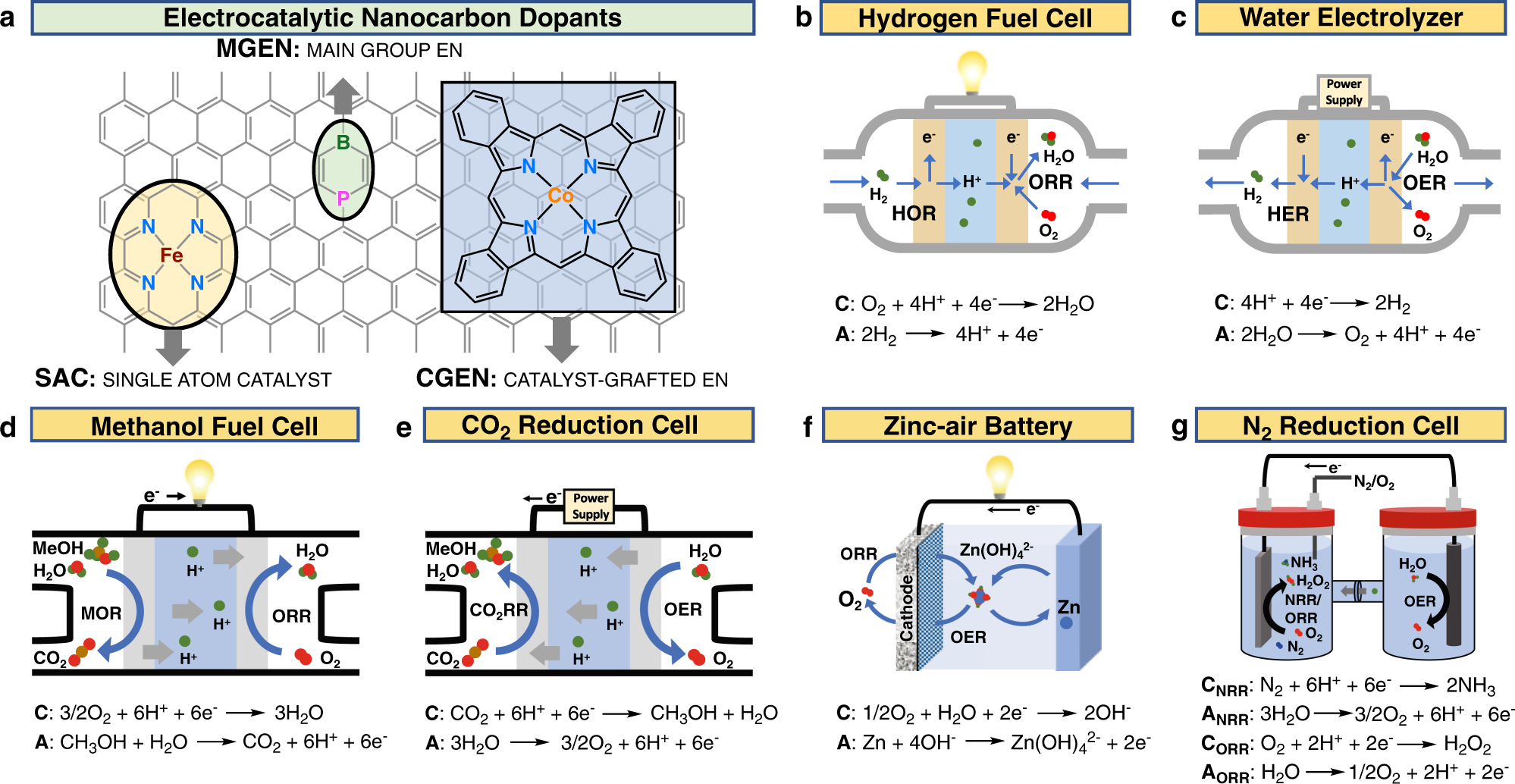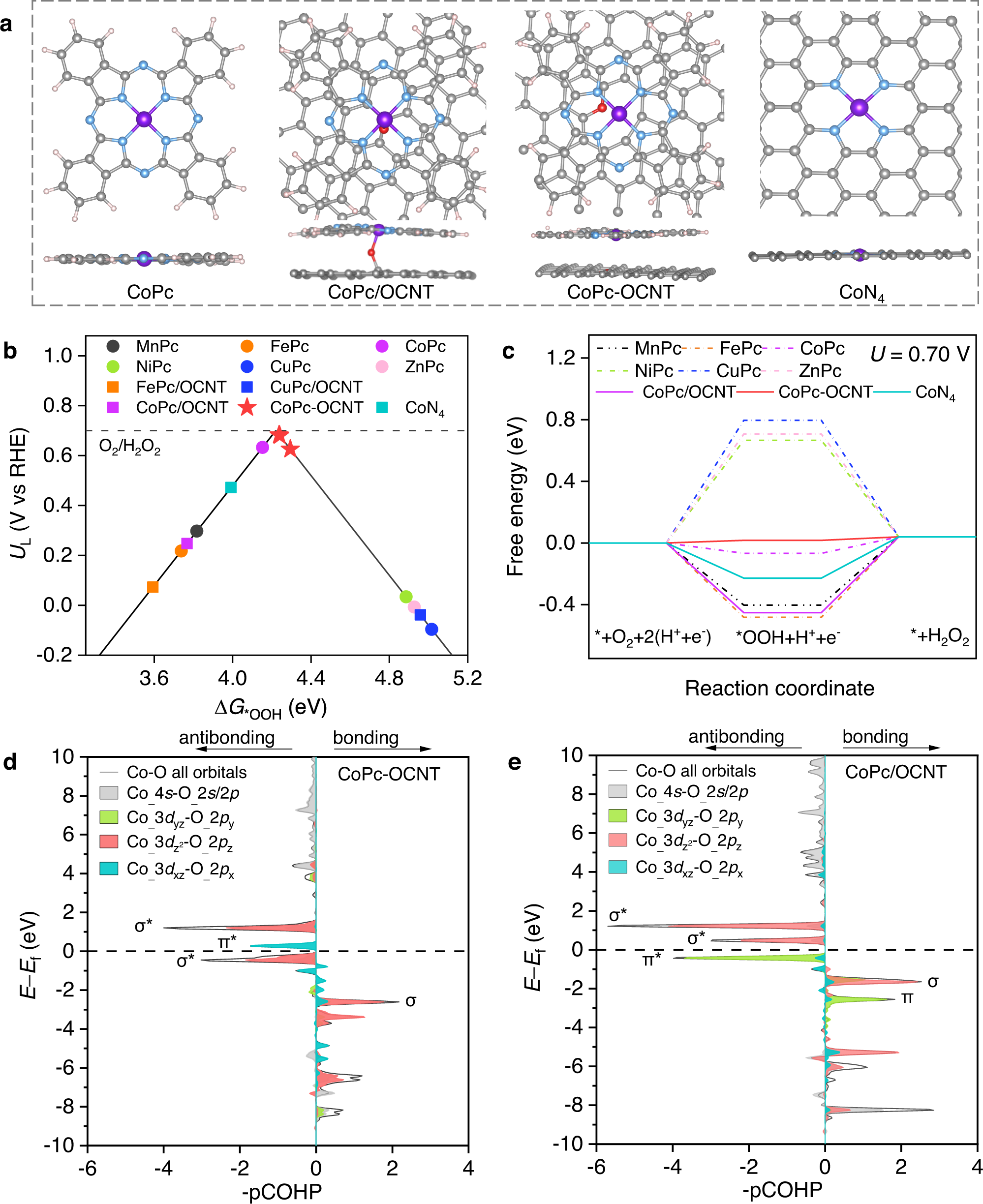
Metal single-site catalyst design for electrocatalytic production of hydrogen peroxide at industrial-relevant currents | Nature Communications
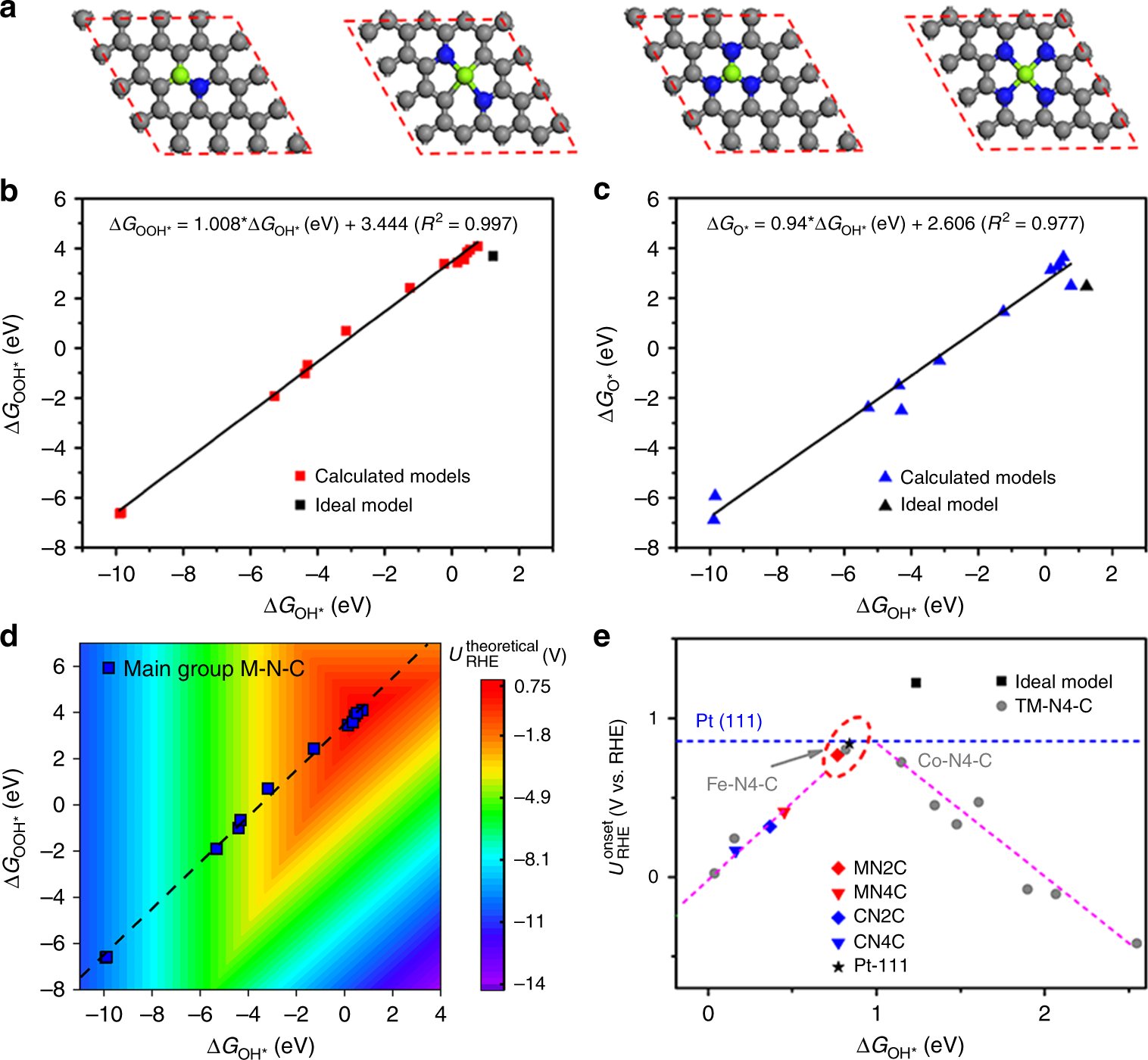
Turning main-group element magnesium into a highly active electrocatalyst for oxygen reduction reaction | Nature Communications
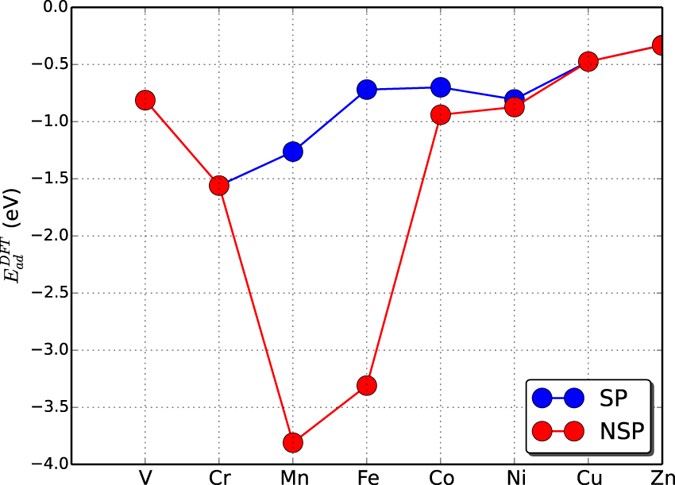
An improved d-band model of the catalytic activity of magnetic transition metal surfaces | Scientific Reports
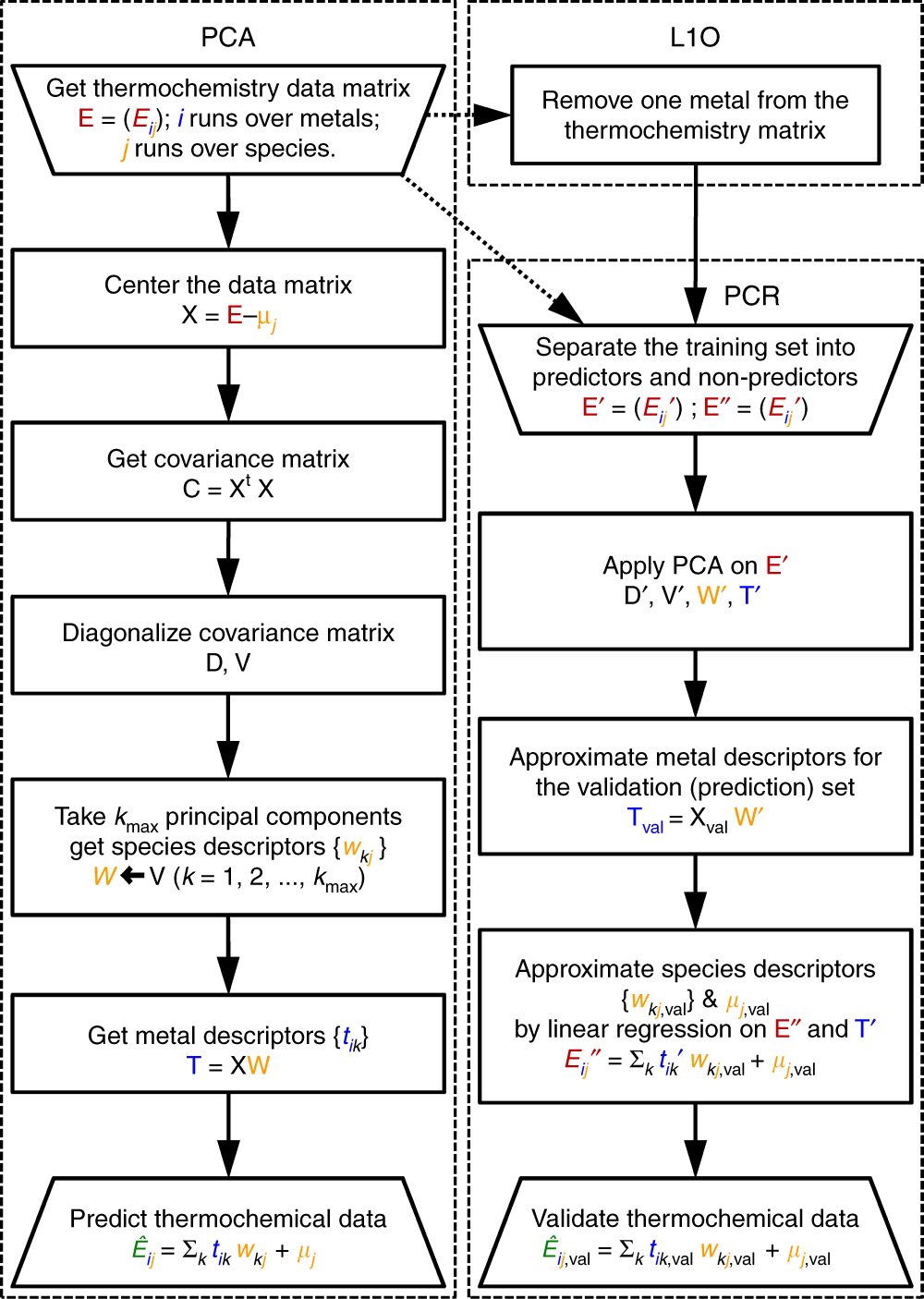
Statistical learning goes beyond the d-band model providing the thermochemistry of adsorbates on transition metals | Nature Communications
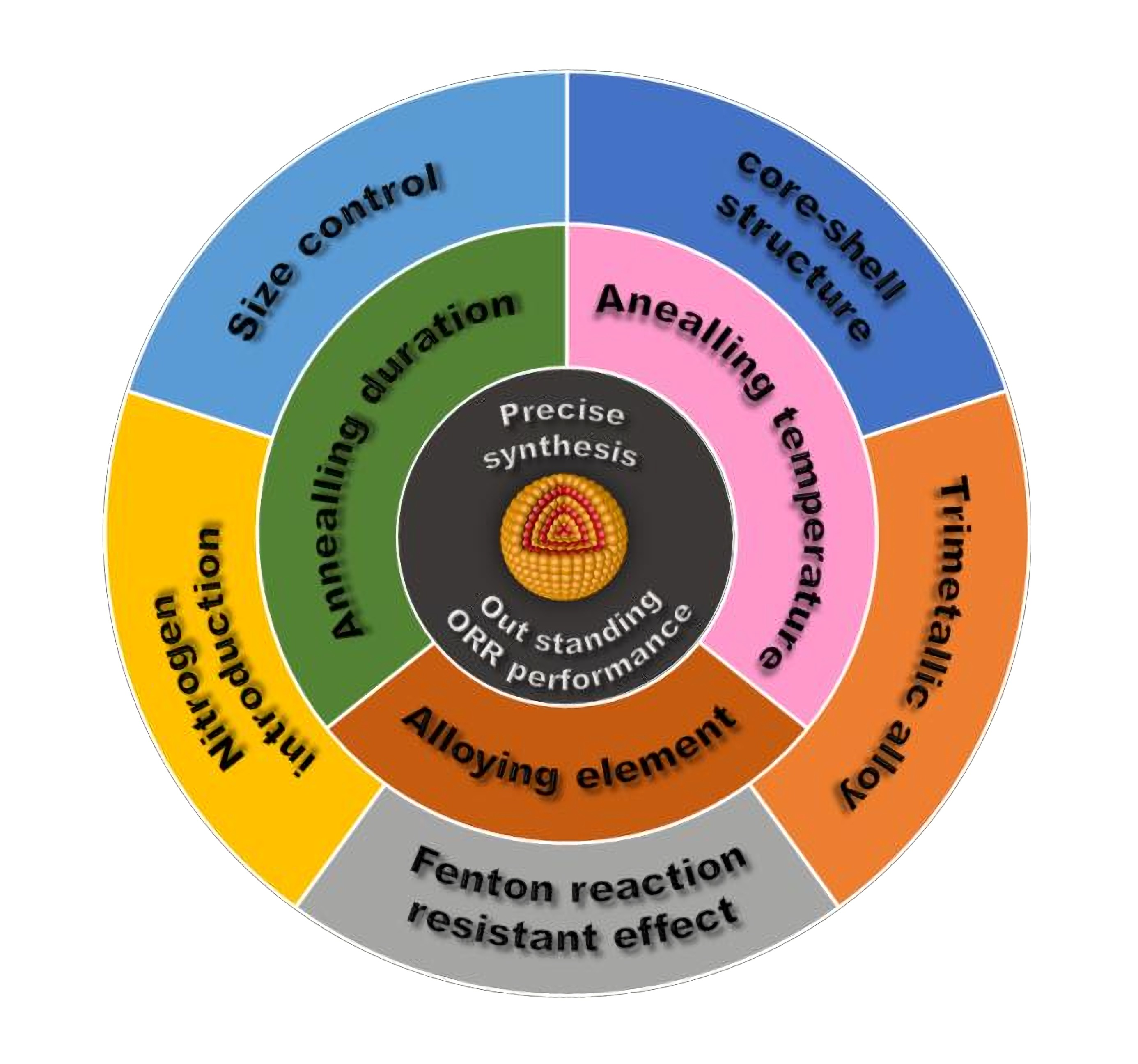
Catalysts | Free Full-Text | Pt-Based Intermetallic Nanocrystals in Cathode Catalysts for Proton Exchange Membrane Fuel Cells: From Precise Synthesis to Oxygen Reduction Reaction Strategy

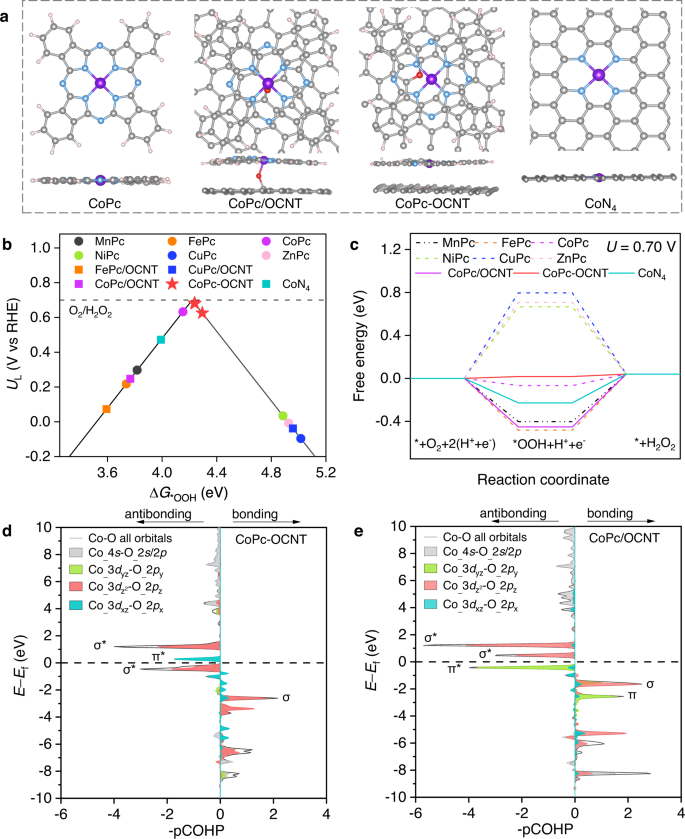
Identifying the impact of the covalent-bonded carbon matrix to FeN4 sites for acidic oxygen reduction | Nature Communications

Measuring and Relating the Electronic Structures of Nonmodel Supported Catalytic Materials to Their Performance | Journal of the American Chemical Society

Pinpointing the axial ligand effect on platinum single-atom-catalyst towards efficient alkaline hydrogen evolution reaction | Nature Communications

A review of oxygen reduction mechanisms for metal-free carbon-based electrocatalysts | npj Computational Materials

Lewis acid–dominated aqueous electrolyte acting as co-catalyst and overcoming N2 activation issues on catalyst surface | PNAS
First principles analysis of hydrogen chemisorption on Pd–Re alloyed overlayers and alloyed surfaces: The Journal of Chemical Physics: Vol 112, No 12
Metal single-site catalyst design for electrocatalytic production of hydrogen peroxide at industrial-relevant currents | Nature Communications
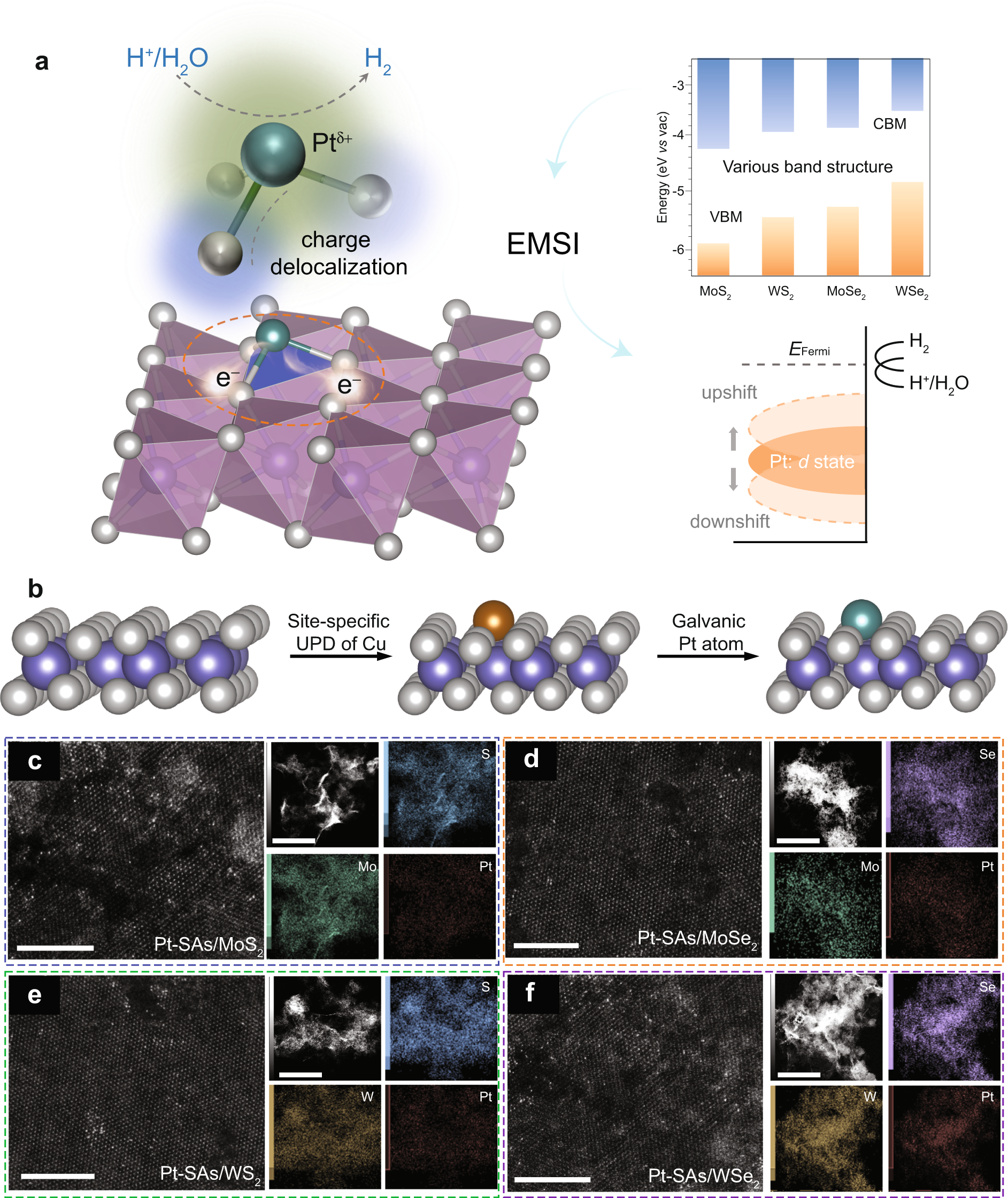
Electronic metal–support interaction modulates single-atom platinum catalysis for hydrogen evolution reaction | Nature Communications

Composition-Tuned (MoWV)Se2 Ternary Alloy Nanosheets as Excellent Hydrogen Evolution Reaction Electrocatalysts | ACS Nano
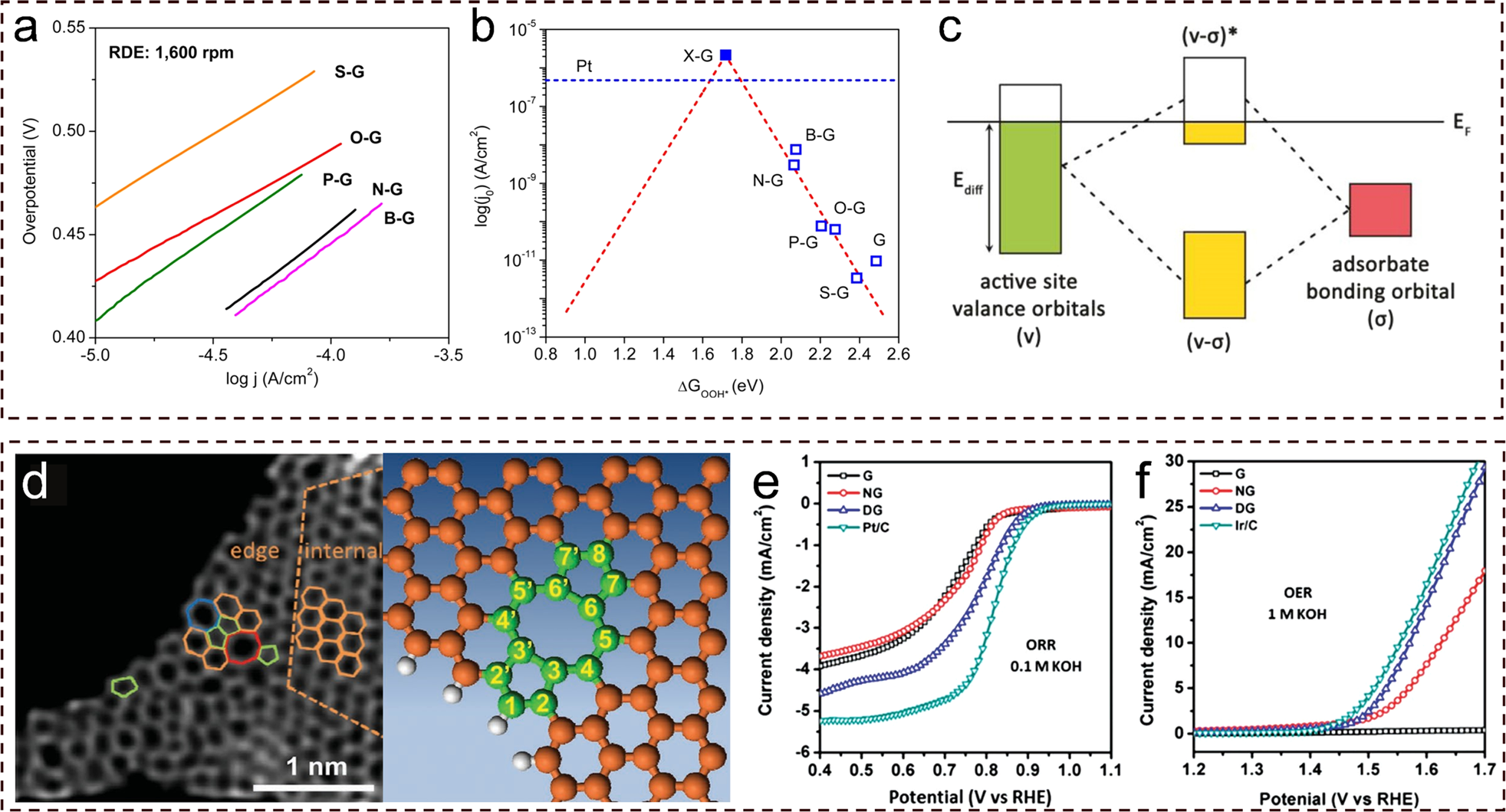
Low-dimensional non-metal catalysts: principles for regulating p-orbital-dominated reactivity | npj Computational Materials
Measuring the Electronic Structure of Alloy Materials and Relating it to their Catalytic Performance

Catalysts | Free Full-Text | Ab Initio Investigation of the Adsorption and Dissociation of O2 on Cu-Skin Cu3Au(111) Surface

Metal single-site catalyst design for electrocatalytic production of hydrogen peroxide at industrial-relevant currents | Nature Communications
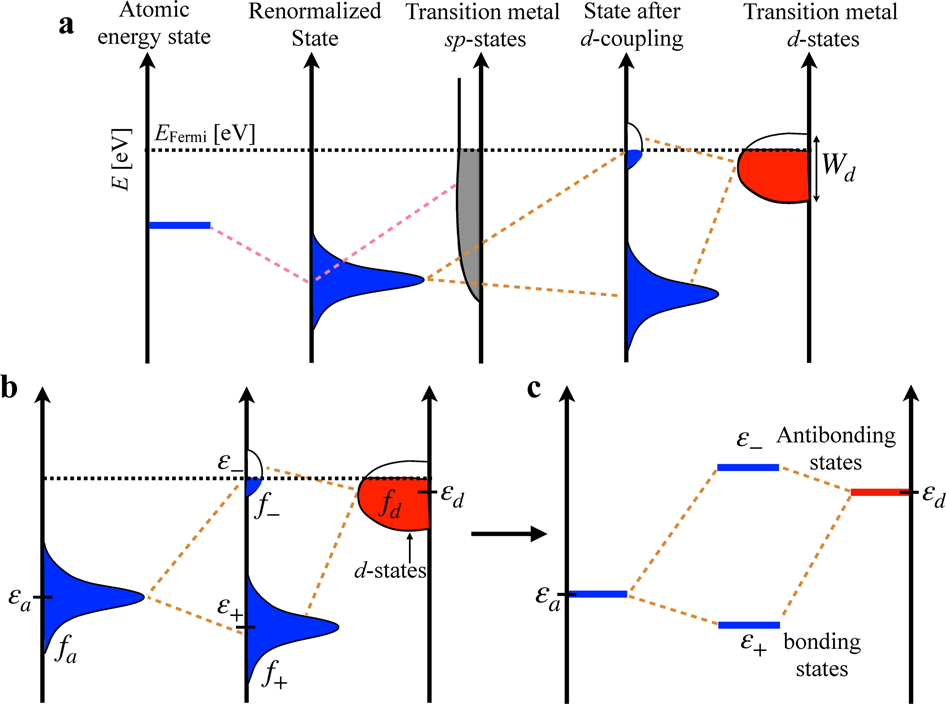
Electronic structure factors and the importance of adsorbate effects in chemisorption on surface alloys | npj Computational Materials

Surface Composition Dependent Ligand Effect in Tuning the Activity of Nickel–Copper Bimetallic Electrocatalysts toward Hydrogen Evolution in Alkaline | Journal of the American Chemical Society

Imaging the facet surface strain state of supported multi-faceted Pt nanoparticles during reaction | Nature Communications

Uncovering electronic and geometric descriptors of chemical activity for metal alloys and oxides using unsupervised machine learning - ScienceDirect